Ionic compounds chloride sodium ions formation definition chemistry Ionic compound ions Explain the formation of ionic bonds with examples
What are Ionic Compounds? - Definition, Structure, Properties
Ionic bond diagram Ionic bond examples Ionic bonding
6.2: comparing ionic and molecular substances
Igcse chemistry 2017: 1.56c: understand why ionic compounds conductWhat are ionic compounds? What are ionic compounds?Ionic compounds examples pictures.
Ionic bonding dot and cross diagramExploring nacl: the ionic compound wonder How does an ionic bond form between sodium and chlorineDifference between ionic and covalent bonds.

Lewis structure of ionic compound
Ionic bond examplesIonic compounds examples and uses Ionic covalent bond vs bonding examples between bonds difference biologyIonic substances.
Edexcel igcse chemistry 复习笔记 1.6 5 ionic compounds: bonds, structureIonic and covalent bonding worksheet Ionic substances (7/12)Ionic compound bond examples bonding example ions compounds ion structure biology nacl chemistry between charged oppositely sodium chloride anion negative.

Ionic formation chemistry
5.7.1 properties of ionic and covalent compounds – revision.myIonic compounds chemistry diagram particles why solids igcse electricity molten conduct solution when aqueous showing only Ionic solid examplesPeriodic table compounds chemistry ionic bonds covalent valence each ions element elements electron family lewis symbols molecular dot has ch150.
2.7: ions and ionic compoundsIonic compound properties (gcse chemistry) What are ionic compounds definition structure propertiesIonic bond: facts, definition, properties, examples, & diagrams.

Ch150: chapter 3 – ions and ionic compounds – chemistry
Magnesium fluoride ionic bonding electron bond mgf2 mgf substances chemie 43ee 649d 9bbb chimieIonic bonding atoms wikipedia sodium naf reaction ions fluorine fluoride Ionic vs covalent which is which and how to tell them apartIonic bonding compounds covalent sodium bonds compound chloride atoms electrons sio2 combine ions example chlorine electron atom nacl magnesium molecules.
Ionic molecular compounds solids substances covalent properties molecules ions interactions comparing bonds bonding representation libretexts metallic methane intermolecular chemDiagram of an ionic compound stock illustration Ionic substances atomic structure level chemistryWhat best describes how an ionic bond forms.

Periodic table with ionic charges and names of elements
Which best compares the properties of ionic and metallic substancesIonic compound chloride sodium compounds chemical conductivity electrical bonds bonding lattice electricity conduct conductive sodio cloruro distilled particles ck dissolved Ionic chemistry atom compounds compound ions chemical molecule vs types between element molecules atoms covalent general principles molecular bonding formulasIonic bonds compounds potassium chloride aplustopper.
Ionic compounds ions formulas polyatomicPhysical properties of ionic compounds ( read ) Ionic compounds spm kimia covalent nacl natriumchlorid padat kristallgitter sifat compound solid gitterstruktur joner struktur ions negativeIonic bond and ionic bond formation, definition, properties in.

Ionic compounds compound cscl nacl magnesium diamond edurev graphite
Ionic bonds definition chemical diagramsIonic compounds fluorite dictionary What are ionic compounds?.
.

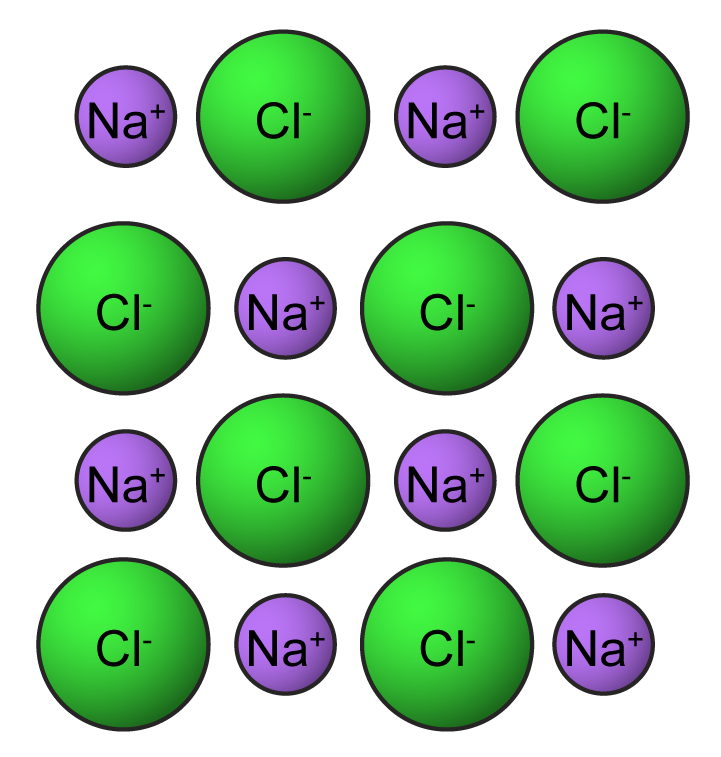
5.7.1 Properties of Ionic and Covalent Compounds – Revision.my

What are Ionic Compounds? - Definition, Structure, Properties

Periodic Table With Ionic Charges And Names Of Elements | Brokeasshome.com

Ionic Bond and Ionic Bond Formation, Definition, Properties in
.PNG)
How Does An Ionic Bond Form Between Sodium And Chlorine - slideshare

Which Best Compares the Properties of Ionic and Metallic Substances
